ESMO Showcases The Aurora Suite™ Approach
Our successful poster submission to the European Society of Medical Oncology (ESMO) Congress highlights how the Aurora Suite™ focuses on science, key data, and actionable insights to optimize trial strategy.
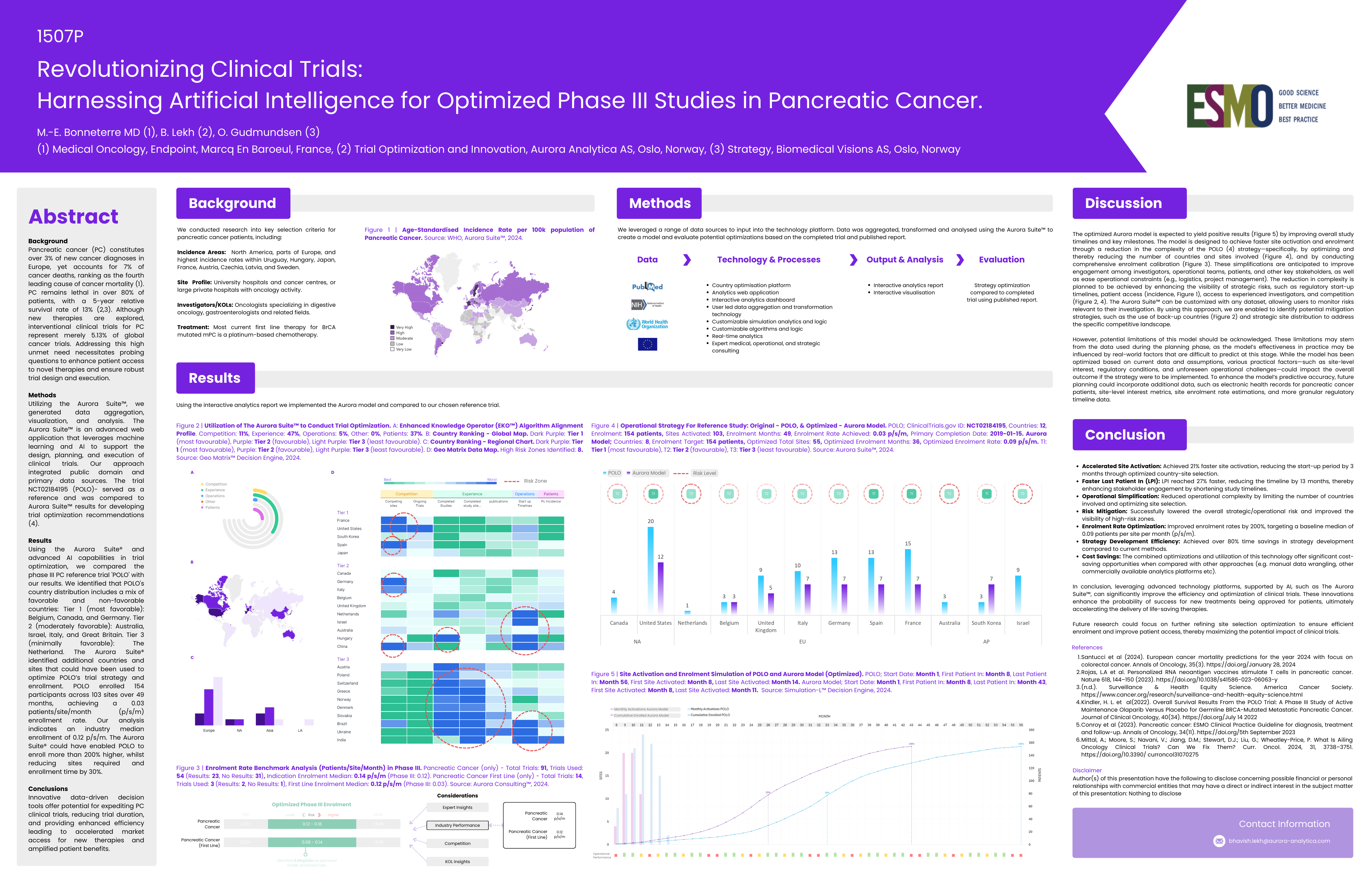
Revolutionizing Clinical Trials: Harnessing Artificial Intelligence for Optimized Phase III Studies in Pancreatic Cancer.
M.-E. Bonneterre MD (1), B. Lekh (2), O. Gudmundsen (3)
Background
Pancreatic cancer (PC) constitutes over 3% of new cancer diagnoses in Europe, yet accounts for 7% of cancer deaths. PC remains lethal in over 80% of patients, with a 5-year relative survival rate of 13%.
Results
POLO enrolled 154 participants across 103 sites over 49 months, achieving a 0.03 patients/site/month enrollment rate. Our analysis indicates an industry median enrollment of 0.12 p/s/m. The Aurora Suite® could have enabled POLO to enroll more than 200% higher, whilst reducing sites required and enrollment time by 30%.
Conclusions
- Accelerated Site Activation: 21% faster site activation, reducing the start-up period by 3 months.
- Faster Last Patient In: LPI reached 27% faster, reducing the timeline by 13 months.
- Enrolment Rate Optimization: Improved enrolment rates by 200%.
- Strategy Development Efficiency: Over 80% time savings in strategy development.